Potassium nitrate (KNO 3), also known as saltpeter or nitre, is used in fertilizers, match heads and pyrotechnics.Įstimated Crustal Abundance: 2.09×10 4 milligrams per kilogramĮstimated Oceanic Abundance: 3. Electron Configuration of the elements Aluminum, Ne3s23p Mercury, Xe6s24f145d Americium, Rn7s25f Molybdenum, Kr5s14d Antimony, Kr5s24d105p. elements, their atomic numbers, symbols, atomic weights, and electron configurations. When we write the configuration we'll put all 19 electrons in orbitals around the nucleus of the Potassium atom. Potassium is in group I so it has 1 unpaired valence electron. :max_bytes(150000):strip_icc()/potassiumatom-58b602485f9b5860464c55ea.jpg)
It is used in respiratory equipment and is produced by burning potassium metal in dry air. In order to write the Potassium electron configuration we first need to know the number of electrons for the K atom (there are 19 electrons). Give the actual electron configuration of each element in the table. It can be found bound to other elements in seawater and minerals, and is needed for plant growth. It reacts violently with water and can even catch fire spontaneously in water. Potassium, the second lightest metal, was discovered by Sir Humphry Davy in 1807. Would you expect to observe a photoelectric effect for potassium using a photon of blue light ( 485 nm). Electron configuration: Ar4s1 Oxidation state: 1 Crystal structure: cubic body centered. Potassium have 2 valence electrons around the nucleus and the atomic number is 19. Potassium superoxide (KO 2) can create oxygen from water vapor (H 2O) and carbon dioxide (CO 2) through the following reaction: 2KO 2 + H 2O + 2CO 2 => 2KHCO 3 + O 2. Removing an electron from solid potassium requires 222 kJ/mol. Potassium is the FIRST element in the FOURTH row of the table.So its the same as Argon, but with one extra electron in its 4s subshell.Check me out: http://. The complete electron configuration of Potassium is 1s2 2s 2 2p6 3s2 3p6 4s1. Potassium carbonate (KHCO 3), also known as pearl ash, is used to make some types of glass and soaps and is obtained commercially as a byproduct of the production of ammonia. Potassium hydroxide (KOH) is used to make soaps, detergents and drain cleaners. By knowing the electron configuration of an element, we can predict and explain a great deal of its chemistry. It is used in fertilizers, as a salt substitute and to produce other chemicals. The electron configuration of an element is the arrangement of its electrons in its atomic orbitals. Potassium chloride (KCl) is the most common potassium compound. Potassium forms many important compounds. There are deposits of billions of tonnes of potassium chloride. 
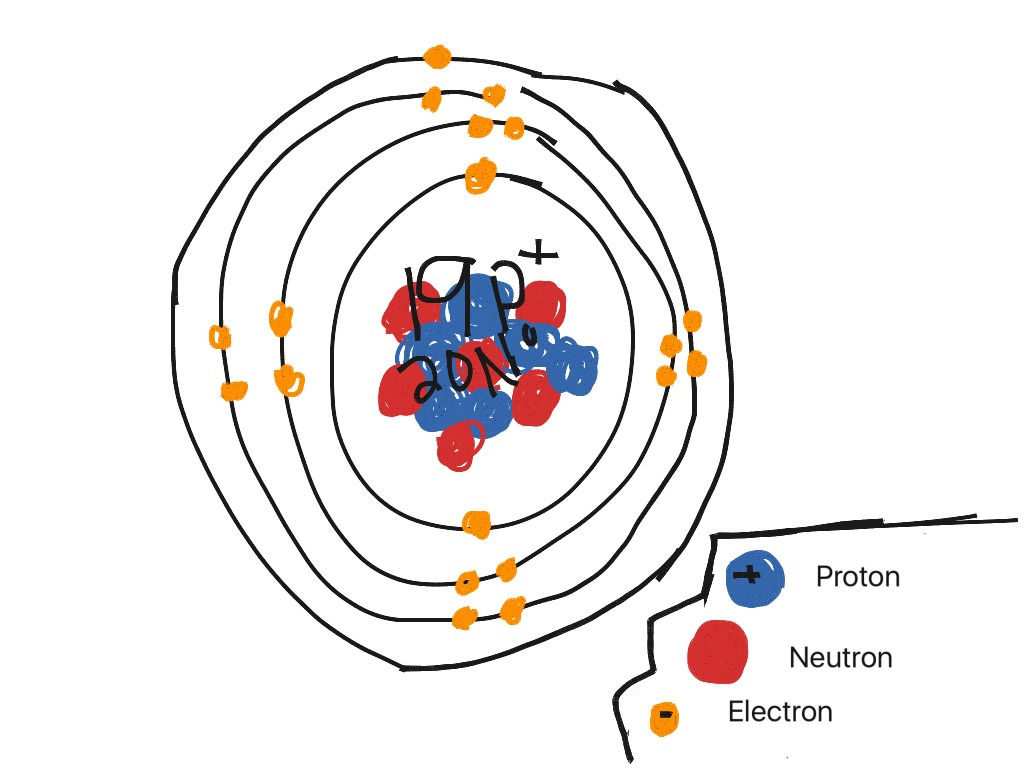
The valence shells of the inner transition elements consist of the ( n 2) f, the ( n 1) d, and the ns subshells. Potassium is the seventh most abundant metal in the Earths crust. They are shown in green in Figure 8.3.6 8.3. Potassium forms an alloy with sodium (NaK) that is used as a heat transfer medium in some types of nuclear reactors. Inner transition elements are metallic elements in which the last electron added occupies an f orbital. To prevent it from reacting with the oxygen and water in the air, samples of metallic potassium are usually stored submerged in mineral oil. Enough heat is produced to ignite the hydrogen gas. Atomic number The number of protons in an atom. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). It reacts with oxygen to form potassium superoxide (KO 2) and with water to form potassium hydroxide (KOH), hydrogen gas and heat. Elements are organised into blocks by the orbital type in which the outer electrons are found. And the electron configuration of potassium is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 1. Pure potassium is a soft, waxy metal that can be easily cut with a knife. Hence the electron arrangement in potassium is 2, 8, 8, 1. Caustic potash, another important source of potassium, is primarily mined in Germany, New Mexico, California and Utah. These minerals are often found in ancient lake and sea beds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
